The Three-Dimensional Periodic Table of George Gamow
![]()
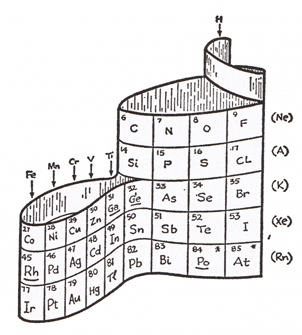
Gamow was a greatly respected scientist, and making scientific concepts clear for the general public was important to him, resulting in the Mr. Tompkins series of science books for the layman in addition to his other scientific works.Clearly, by the style of the drawings, it is clear he felt the concept worthy of a patent application. The section of Mr. Tompkins Gets Serious containing the periodic table drawing above is from The Atom and its Nucleus, published in 1961, so it is not possible that he applied for and was prevented from being awarded a patent by the Alexander Arrangement having been prior art, a common reason for rejection, as my 3D periodic table was invented in 1965, my patent application was only filed in 1969 – after his death in ’68. It could be that his examiner was better informed than I and mine about Courtines and de Chancourtois, our now identified precursors.Finding the original Mendeleev periodic table “shaky”, and apparently feeling that the flat descendents would not be best for introduction of the periodic table, he chose to create one that would serve better, writing; “It is convenient to represent the periodic table of elements by a three-dimensional spiral structure”, which his drawing shows avoids the usual gaps and discontinuities.
He goes on to describe it as “...the backbone of the structure is the column containing the noble gases running all the way from He down to Rn. The next column to the right contains the alkali metals, with Hydrogen placed at the top because its chemical properties are similar to those of the alkali metals. To the left and around the corner from the noble gas column is the one containing the halogens.” Here he surprises us by making clear that his noble gases begin the periods!
Gamow continues to describe the illustrations – fig. 1-2; “The first two periods, from H to F and from Ne to Cl, contain 8 elements each and fall neatly into this scheme, but the next period contains 18 elements and constitutes a problem.”
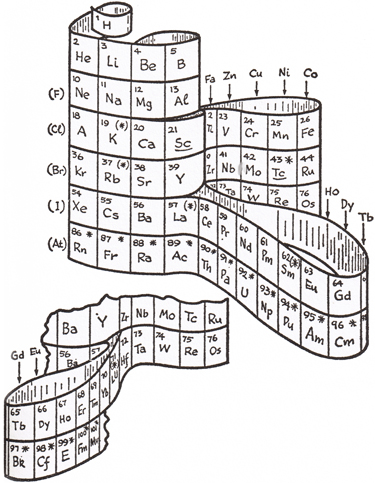
Here he begins to identify the reasons for using a 3-D form instead of the de-facto table; “On the basis of chemical properties, there seems to be no doubt that the 3 elements that follow A (K, Ca, and Sc) must be placed under the 3 corresponding elements (Na, Mg, and Al) of the preceding period and that those preceding Kr (As, Se, and Br) should be under those preceding A (P S, and Cl), but we do not seem to have places for the remaining 11 elements (Ti to Ge).”
Continuing to describe a ribbon of elements; “To dispose of this difficulty,” he continues, “we place Ti and Ge, which both resemble Si, under that element and make an extra loop to accommodate the remaining 9 elements (V to Ga)." Gamow notes that the “same situation arises in the next and in all of the following periods so that the extra loop perpetuates itself all the way to the end of the known sequence of elements.”
Addressing the f-block now, he says; “In the beginning of the fifth period we encounter further trouble of the same kind and are forced to build another extra loop to accommodate the 14 elements (Ce to Lu), known as the rare earths.”
He imagines that Mendeleev “would probably be horrified”, but judges that this 3-D form “gives a very good representation of properties of the different elements.”
His drawing caption says; “The periodic system of the elements represented as a wound ribbon”, and identifies the indicators of radioactive elements, and those elements which were predicted by Mendeleev.
< BACK
![]()
![]()